In mammalian cells, both normal cells and malignant proliferating cells need to obtain essential amino acids (EAAs) to maintain normal life activities. Essential amino acids (methionine, valine, lysine, isoleucine, phenylalanine, leucine, threonine, histidine) are not only proteins, lipids, nucleic acids and other biological macromolecules The synthesis provides "raw materials" and can also act as a signal molecule to induce the activation of the mTOR pathway.
Compared with normal cells, tumor cells often show the important feature of taking up a large amount of essential amino acids. However, the molecular mechanism of the abnormal metabolism of essential amino acids is still unclear.
On December 26, the research group of Professor Qing Guoliang from the Medical Research Institute of Wuhan University published online research results titled "Oncogenic MYC Activates a Feedforward Regulatory Loop Promoting Essential Amino Acid Metabolism and Tumorigenesis" in Cell Reports. The research reported on Myc -SLC7A5/SLC43A1 positive feedback loop reprograms the molecular mechanism of essential amino acid metabolism to promote the malignant development of tumors. This study not only reveals a new mechanism by which metabolic abnormalities promote tumorigenesis and development, but also provides a potential target for the treatment of patients with tumors with high MYC expression.
MYC is a key protein in the regulation of tumor metabolism. In this study, the researchers first proved that the oncoprotein MYC is a key transcriptional regulator that activates the uptake of essential amino acids by tumor cells. So how is MYC regulated?
Many studies in the past have shown that many SLC family member proteins (such as SLC7A5/A8, SLC43A1/A2, SLC6A14, SLCA1-A11 and other amino acid transporters) are involved in regulating the intake of essential amino acids. With the above information, it is obvious to ask whether MYC regulates the above-mentioned SLC family proteins. Further research found that MYC regulates the expression of SLC7A5 and SLC43A1 through direct activation, and forms a MYC-SLC7A5/SLC43A1 positive feedback loop, which enhances the uptake of essential amino acids by tumor cells and inhibits the GCN2-eIF2α-ATF4 stress pathway. Then it selectively activates the protein translation of key cancer-promoting genes such as Myc, Bcl2, Cyclin D1, and ultimately promotes the malignant development of tumors (below).
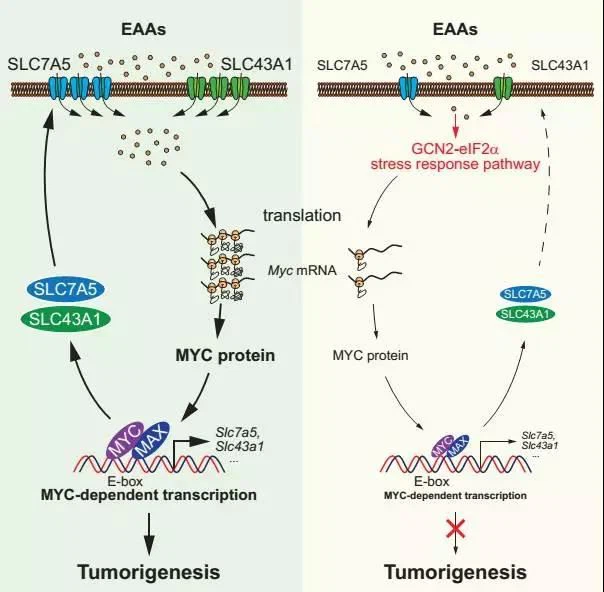
The continuous activation of this loop not only ensures the intake of essential amino acids, but also cascades the entire transcriptional regulatory network mediated by Myc to promote the metabolic reprogramming of key nutrients such as glucose, glutamine, nucleotides, and fatty acids. Interfering with the function of SLC7A5/SLC43A1 can interrupt this positive feedback signal loop, cause the expression of Myc, Bcl2, and Cyclin D1 to decrease in tumor models in vivo and in vitro, and selectively induce apoptosis of tumor cells overexpressing Myc. The above results suggest that the monoclonal antibody targeting SLC7A5/SLC43A1 can be used as a potential treatment for patients with tumors with high MYC expression.
It is reported that Professor Qing Guoliang and Professor Liu Hudan of the Medical Research Institute of Wuhan University are the corresponding authors of the paper, and the doctoral student Yue Ming is the first author of the paper.